 Paul Offit, a member of the FDA’s vaccine advisory committee, during an appearance on CNN’s “New Day” on Tuesday. “They appear to be roughly equivalent,” said Dr. The two vaccines are very similar, but they have a few key differences that make Moderna’s vaccine “more flexible,” as US Health and Human Services Secretary Alex Azar said last month.Įfficacy: Both Moderna’s and the Pfizer-BioNTech vaccine have shown similar efficacy levels of near 95%. How is Moderna’s vaccine different from Pfizer’s? The vaccine also contains fatty materials called lipids, tromethamine, tromethamine hydrochloride, acetic acid, sodium acetate and sucrose. The Moderna vaccine in particular contains a synthetic mRNA that codes for a structure called the “pre-fusion stabilized spike glycoprotein” of the virus. If a vaccinated person is later exposed to the coronavirus, those antibodies should stand at the ready to attack the virus. Once injected, the body’s immune system makes antibodies to the spikes. Like Pfizer’s vaccine, Moderna’s delivers messenger RNA, or mRNA, which is a genetic recipe for making a piece of the spikes that sit atop the coronavirus. About 10% of study participants were Black and 20% were of Hispanic or Latino. The vaccine was effective across age groups, genders, racial and ethnic groups, and participants with medical comorbidities associated with high risk of severe Covid-19, according to the briefing document. One of them died.Īnother 15,000 participants were given the vaccine, and only 11 of them developed Covid-19. 
Over several months, 185 of them developed Covid-19, with 30 developing severe forms of the disease. In Moderna’s trial, 15,000 study participants were given a placebo, which is a shot of saline that has no effect. 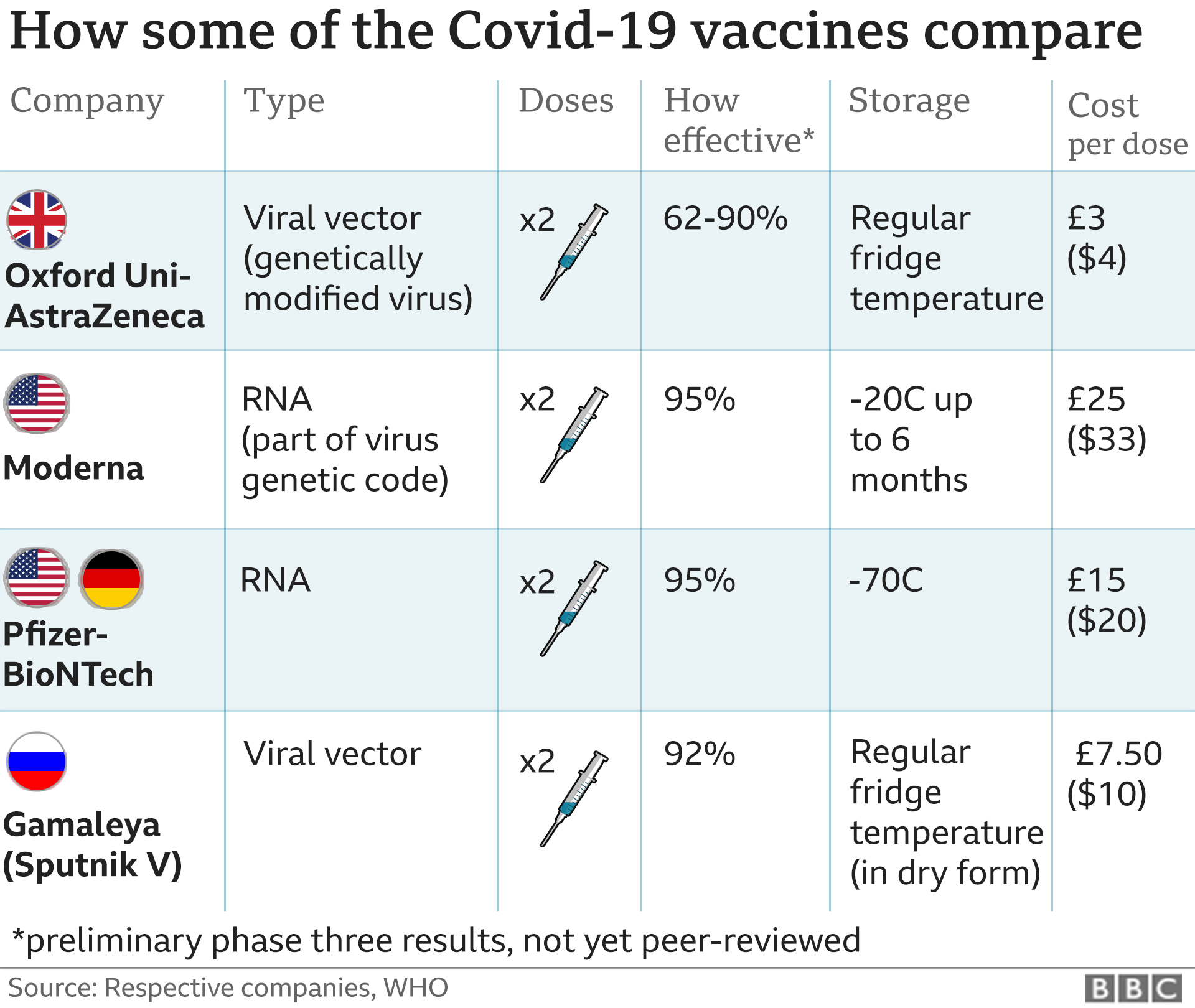 
Here's a look at how the different coronavirus vaccines work The vaccine’s efficacy against Covid-19 was 94.1%, occurring at least 14 days after the second dose, according to a briefing document released by an advisory committee to the FDA.Ĭhina coronavirus covid 19 experimental vaccines development Watson pkg intl ldn vpx_00001308.jpg The Moderna vaccine was effective in preventing Covid-19 cases in total and in limiting severe Covid-19 during the trial. The company applied to the FDA for emergency use authorization for the vaccine on November 30. 
Its advanced stage clinical trial started July 27 and it was the first government-funded Phase 3 clinical trial for a Covid-19 vaccine in the US. The FDA green-lit clinical trials of Moderna’s vaccine on March 3, the first out of the gate. The US Biomedical Advance Research and Development Authority, known as BARDA, supported late-stage clinical trials and has helped scale up manufacturing. The vaccine, called mRNA-1273, had development help from the National Institute of Allergy and Infectious Diseases. When Chinese researchers released the genomic sequence for the novel coronavirus in mid-January, they had a jump start. The company’s scientists had already been collaborating with researchers from the National Institutes of Health on a vaccine for another coronavirus, Middle Eastern Respiratory Syndrome (MERS). It had also never brought a product to phase three of a clinical trial previously. While Pfizer is a pharmaceutical behemoth, Moderna – short for modified RNA – is a biotech company based in Cambridge, Massachusetts.Įstablished in 2010, Moderna has never brought a product to market, or gotten any of its nine or so vaccine candidates approved for use by the FDA. Moderna headquarters in Cambridge, Massachusetts. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
